German team overcomes two perovskite QD hurdles
Perovskite quantum dots are promising materials for LEDs, photocatalysis, and future quantum light sources. Now researchers at Ludwig-Maximilians-University (LMU) in Munich have managed to master two major hurdles in terms of their stability in solution and precise control of their growth.
The team reported the results in the Journal of the American Chemical Society and in ACS Energy Letters in a paper titled ''Polar Opposites: Ligand-Mediated Polarity Inversion for Perovskite Quantum Dots with Sub-Nanometer Ligand Shells'.
Although perovskite quantum dots are comparatively easy to manufacture in solution, their soft ionic crystal lattices make them sensitive to many solvents. Particularly problematic are polar solvents like alcohols, in which quantum dots often disintegrate quite rapidly.
“A challenge to date has been keeping the quantum dots stable without impairing their structural and optical properties,” says Quinten Akkerman from the Nano-Institute Munich and the Faculty of Physics at LMU. Together with his team, Akkerman has developed a strategy to get around these limitations.
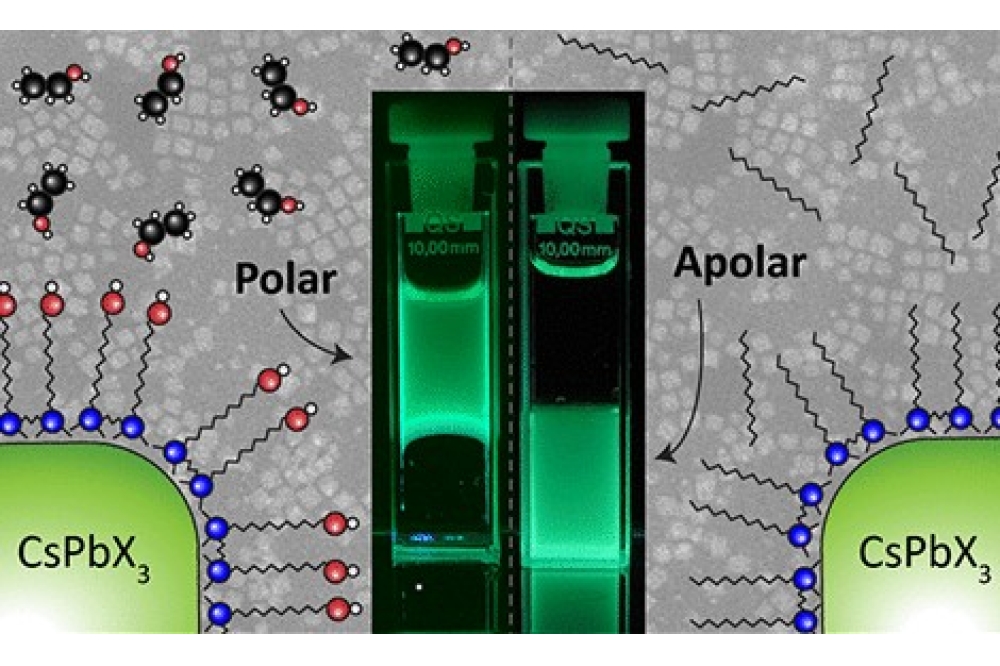
The scientists used so-called Gemini ligands, which form a stable molecular shell around the quantum dots. They bind with their charged groups to the surface of the quantum dots, while at the same time their structure forms a polar external surface. This allows the quantum dots to disperse in a stable manner in polar solvents like ethanol. The ligand layer remains exceptionally thin at around 0.7 nm, such that the optical properties of the quantum dots are preserved.
The stabilised quantum dots continue to exhibit high photoluminescence quantum yields and remain preserved in solution for long periods of time. At the same time, they can now be processed in so-called green solvents – an advantage for future manufacturing processes in optoelectronics.
In a second study, the team addressed the question of how to precisely control the size and structure of perovskite quantum dots. These properties determine what colour and intensity the quantum dots emit.
Akkerman’s team developed a method by which the formation of new seed crystals is specifically suppressed. Instead, existing quantum dots grow in a controlled manner. This becomes possible through precise coordination of the reaction conditions and the ligands employed, which influence the reaction kinetics.
With a multi-stage injection strategy, the researchers were able to control the growth of the quantum dots over longer periods. They even achieved sub-unit-cell precision – that is to say, with a precision smaller than an individual crystal lattice cell.
The quantum dots generated in this fashion exhibit particularly narrow size distribution and stable optical properties. Such controlled structures are an important precondition for use in LEDs or future quantum light applications.
“Together, the two studies provide new approaches for solving challenges relating to perovskite quantum dots,” says Akkerman. “While the new ligand chemistry improves their processing and stability, the precise control of their growth enables precise tuning of their optical properties.” This opens up new possibilities for applications in optoelectronics and future quantum light technologies.